Statsols looked at data from 220,000 clinical trials. Below are just some of the points we found.
As you may know, Section 801 of the Food and Drug Administration Amendments Act (FDAAA 801) requires the registration and submission of summary results of clinical trials with ClinicalTrials.gov. The law applies to certain clinical trials of drugs (including biological products) and medical devices. At the time of publishing ClinicalTrials.gov currently has over 220,000 clinical trials registered. We examined the data to get some interesting results on a few topics such as:
- Where in America are the most clinical trials registered
- How many clinical trials were registered in 2015
- How many trials were registered per week
- What is the busiest time for clinical trial submission
- What sample size calculator did CROs and Pharmaceutical companies use who received FDA approval for their clinical trials
Clinical Trials Data Dive - Trends from 220,000 Clinical Trials
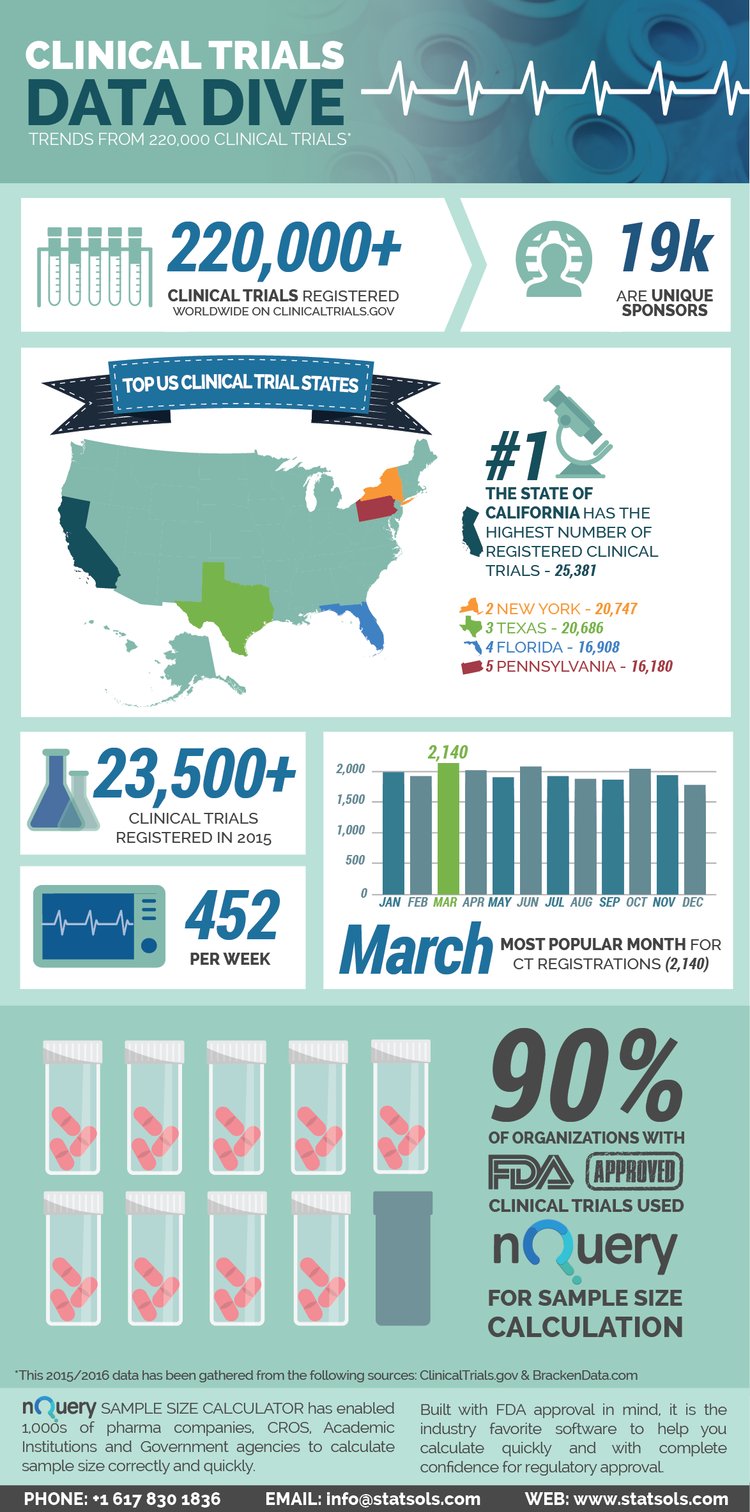
What do you think of the findings? Will New York overtake California as having the highest number of clinical trials registered? Let us know below.
We will be posting more findings soon so subscribe or follow us on LinkedIn and Twitter.
About Statsols
Statsols provide nQuery Sample Size Calculator. Used by 45/50 top pharma companies, it is the industry favorite choice for easily calculating complex sample size for successful clinical trials.
Share this Image On Your Site - Just copy and paste the code below.

















No Comments Yet
Let us know what you think