Why do Phase III clinical trials fail and how can we improve them? Dr. Grignolo, PhD, Corporate Vice President of PAREXEL International, the multinational life sciences consulting firm, recently examined this issue in a paper titled: Phase III Trial Failures: Costly, But Preventable.
We have reviewed this journal and brought you the primary findings as they appeared in Applied Clinical Trials Volume 25, Issue 8 and also on appliedclinicaltrialsonline.com.
Why do phase III clinical trials fail?
Dr. Alberto Grignolo of Parexel undertook a study to answer this question.
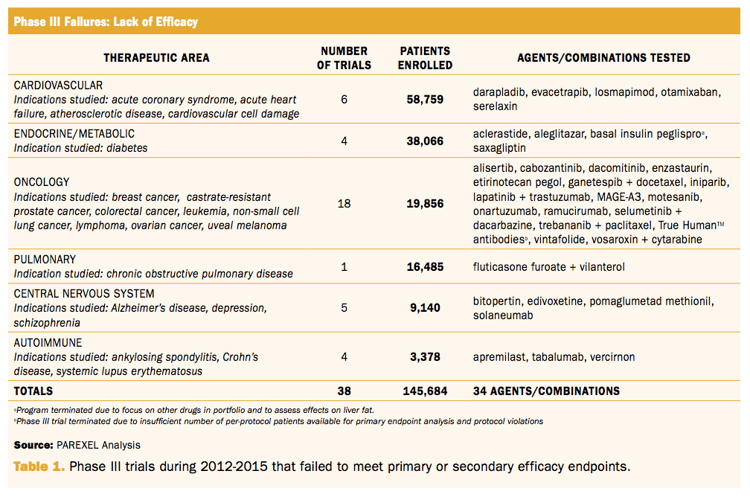
 Grignolo and his researchers selected 38 failed Phase III trials spanning mid 2012 to 2015, where the data was publicly available and then evaluated these in a variety of methods to see what data and lessons could be extracted. It is worth noting that the 38 phase III trials that failed, had a significant enrollment total of 150,000 patients.
Grignolo and his researchers selected 38 failed Phase III trials spanning mid 2012 to 2015, where the data was publicly available and then evaluated these in a variety of methods to see what data and lessons could be extracted. It is worth noting that the 38 phase III trials that failed, had a significant enrollment total of 150,000 patients.
Previous studies have examined this important and costly question before. Dr. Grignolo's paper first references a previous study by the Tufts Center for the Study of Drug Development (CSDD) evaluating clinical trials from 2000 to 2009. This previous study concluded that the three most common reasons for failure in Phase III development is:
- Lack of Efficacy — i.e., failure to meet the primary efficacy endpoint
- Lack of Safety — i.e., unexpected adverse or serious adverse events
- Lack of Commercial / financial benefit— i.e., failure to demonstrate value compared to an existing therapy
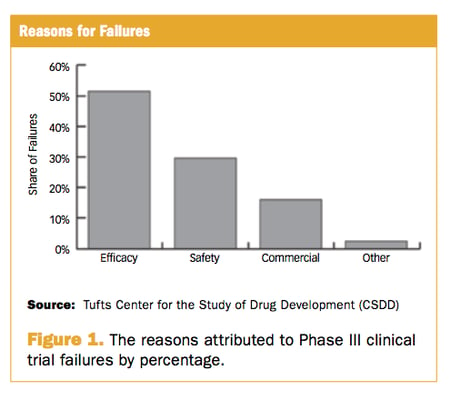
Parexel expanded on this research and conducted their own analysis into phase III failures due to efficacy. The results of their findings are listed in the table below:
So after establishing efficacy as the primary driving factor for phase III clinical trial failures Dr. Grignolo then examines the failures by a number of criteria including new molecular entities, therapeutic area and molecule type.
The rest of the report is split into six parts. To go directly to a particular entity just click on the name below. Otherwise, please read on.
- Phase III clinical trial failures of new molecular entities
- Phase III clinical trial failure rates by therapeutic area
- Phase III clinical trial failure rates by molecule type
- Phase III clinical trial failure due to insufficient sample size
- How to reduce phase III clinical trial failure
- What future strategies may we see to prevent Phase III failures
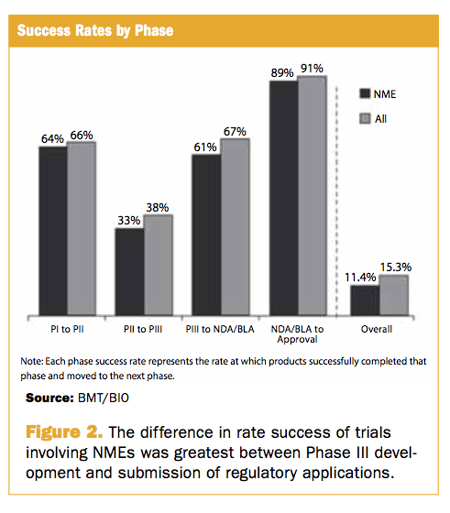
1: Phase III clinical trial failures of new molecular entities (NME)
A study by BioMedTracker (BMT) and the Biotechnology Industry Organization (BIO) evaluated R&D projects involving more than 9,500 different drug and biological products from 2004 to 2014.
- In each phase analyzed, the failure rates of trials involving NMEs were higher than trials involving all drugs, and the difference was greatest between Phase III development and submission of regulatory applications (See Figure 2)
- Among the Phase III trials analyzed, only 61% of NME trials succeeded in moving to the application phase (39% failure rate)
- Whereas 67% of all drug trials moved to the application phase (33% failure rate)
The high failure rate of Phase II trials reported in that analysis (62% and 67%, respectively) is not unexpected for exploratory trials.
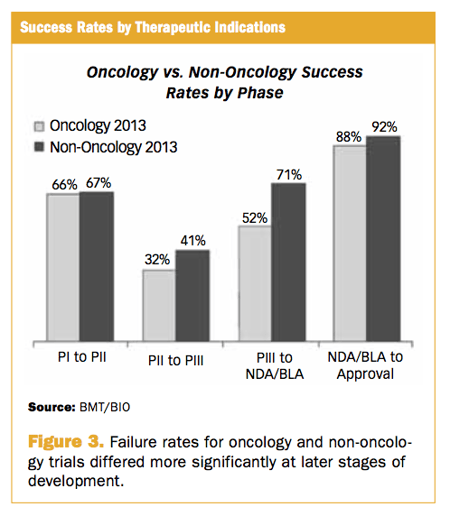
2: Phase III clinical trial failure rates by therapeutic area
Among the Phase III trials evaluated, not surprisingly oncology and cardiovascular trials had the highest failure rates (data not shown).
The higher failure rate for oncology trials might be due to the inclusion of survival endpoints and the need to show efficacy by an improvement in overall survival.
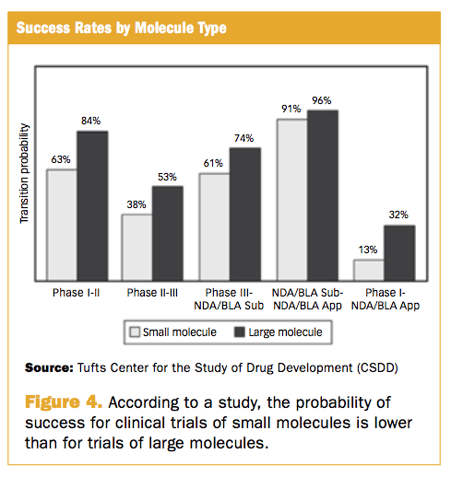
3: Phase III clinical trial failure rates by molecule type
Failure rates differ by type of molecule. A study by the Tufts CSDD found that the probability of success for clinical trials of small molecules is lower than for trials of large molecule.
- Among the Phase III trials, analyzed, only 61% of studies involving small molecules progressed from Phase III testing to the regulatory application phase (39% failure rate)
- Trials of large molecules, however, were more successful in moving to the application phase, with a success rate of 74% (26% failure rate)
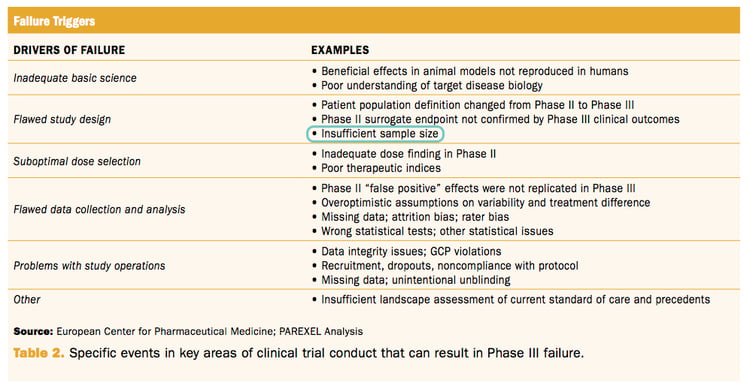
4: Phase III clinical trial failure due to insufficient sample size
In September 2014, The European Center for Pharmaceutical Medicine (ECPM) organized a seminar titled “Why Clinical Trials Fail”. Insufficient sample size was highlighted as a factor that leads to study failure.
However, an insufficient sample size calculation is a risk factor that can be greatly controlled by using nQuery sample size and power calculation software for successful clinical trials.
nQuery is sample size software that is specifically designed for clinical trials and regulatory approval. In 2017, 90% of organizations with FDA approved clinical trials used nQuery for sample size calculation. Below is a brief snap shot of the core tables in the current version of nQuery.

We encourage you to examine our applied example page. This provides all researchers with guided sample size examples in nQuery that are divided by both statistical and therapeutic method. In addition, to increase your understanding of sample size, our Professional Development Sample Size Training is open to both current nQuery users and non nQuery users.
5: How to reduce phase III clinical trial failure?
Confirming the efficacy of a new drug or biologic is not a task that can be simplified, it can be improved though. Due to the nature of Parexel being an international CRO, Grignolo states that
“Given our unique perspective in working with hundreds of companies across thousands of clinical trials and compounds, we and numerous colleagues at PAREXEL are exposed to these approaches on a daily basis”.
The 5 R Framework
In 2011, AstraZeneca aimed to overhaul its R&D process to improve the health of the organization and increase the chance of success of its Phase III trials.
By evaluating its small-molecule drug projects over a 5 year period AstraZeneca identified the factors associated with project success and developed a framework that now drives its development process. These strategies can be implemented during the entire development process, in specific phases of development, and/or during clinical trial design.
The 5R framework guides R&D teams in identifying the right target, the right tissue, the right safety, the right patients and the right commercial potential.

Optimizing Phase III Clinical Trial Design
Flaws in clinical trial design are a major driver of Phase III failures. Several strategies have been developed for optimizing trial design. The next section aims to highlight a few of them:
Review and optimization
- Typically, a number of trade-off decisions need to be made in the compilation of most Phase III protocols—e.g., will the protocol include a specific secondary objective that has commercial value but potentially prolongs the duration of the study? Identifying and quantifying the impact of these trade-offs is helpful in designing better protocols.
Modeling and simulation
- Modeling and simulation is currently being used more broadly than just for the selection of an optimal dose. Modeling and simulation of various clinical trial designs is one such example that is being used in an effort to design optimal Phase III studies.
Adaptive Designs
- Adaptive designs provide an opportunity to assess interim data and to sense-check some of the initial uncertainties or assumptions that were made at the outset of the trial. More importantly, these provide an opportunity to change course during a trial and to correct these incorrect assumptions in a prospectively planned manner that does not jeopardize statistical validity and the operational integrity of the trial.
Biomarkers
- Biomarkers are being used increasingly to assess efficacy in a rapid and more objective manner.
- A particular challenge associated with the use of biomarkers in clinical trials is the requirement to validate these biomarkers as relevant disease-modifying endpoints and correlate changes in these endpoints to clinically significant changes in disease progression.
Enrichment strategies
- Enrichment strategies involving genotyping are being used increasingly to optimize the proposed study population to those individuals most likely to respond to the treatment under investigation.
De-risking study execution through data
- Leveraging data from a variety of sources (e.g., electronic health records [EHR], prior site performance, central labs, etc.)
- Ongoing surveillance of the quality of data being collected during a Phase III trial and tying this to a properly planned risk-based monitoring.
Reducing the risk of insufficient sample size calculation
- In addition to the previous points, we would like to highlight the importance of engaging with your biostatistician as early as possible. The benefits of this include a more efficient study design and greater chance of calculating the correct sample size through sample size software such as nQuery.
6: What future strategies may we see to prevent Phase III failures?
The paper lists examples of what other areas that are currently being explored and could hold potential in reducing Phase III failure. They include:
- Replacement of the current gold standard, the randomized controlled trial
- Wearable devices that collect real-time data
- Adaptive licensing
- Next-generation sequencing and improved understanding of the genetic basis of disease
- Basket/master protocols
CONCLUSION
Dr. Grignolo concludes his publishing with the following
“We believe that the current failure rate in Phase III studies is unacceptably high, and that industry is keen on reducing this failure risk, although some in industry may believe that failure is the price to be paid for innovation.
As a first step, it is important to understand the reasons and root causes driving these failures. Our research identified recently failed Phase III studies that have enrolled nearly 150,000 patients. Based on data from our analysis and others, we have listed the main reasons why Phase III trials fail. In addition, and given our unique perspective in working with hundreds of sponsors across thousands of trials, we have highlighted some of the approaches that pharmaceutical companies are implementing in an effort to reduce these costly late-stage failures.
Along with our colleagues in the pharmaceutical industry, we are optimistic about the potential of some or all of these approaches to improve the Phase III success rate".
The full link to both articles of Phase III Trial Failures: Costly, But Preventable can be accessed here:
http://www.appliedclinicaltrialsonline.com/phase-iii-trial-failures-costly-preventable
https://www.parexel.com/files/5014/7274/5573/ACT_Article.pdf
Follow up Webinar: FDA Guidance on Adaptive Sample Size Re-estimation
Given the high failure rates and the increased costs of clinical trials, researchers need innovative design strategies to best optimize financial resources and reduce the risk to patients.
Adaptive designs are emerging as a way to reduce risk and cost associated with clinical trials. The FDA recently published new draft guidance on adaptive trials and are actively encouraging sponsors to use Adaptive trials.
Adaptive trials are a type of clinical trial design that allows adaptations or modifications to aspects of the trial while it is on-going, without undermining the validity and integrity of the trial. One common adaption is a sample size re-estimation, which increases the sample size in response to interim data.

















 Grignolo and his researchers
Grignolo and his researchers 








No Comments Yet
Let us know what you think